|
12/9/2023 0 Comments Hcn molecular geometry 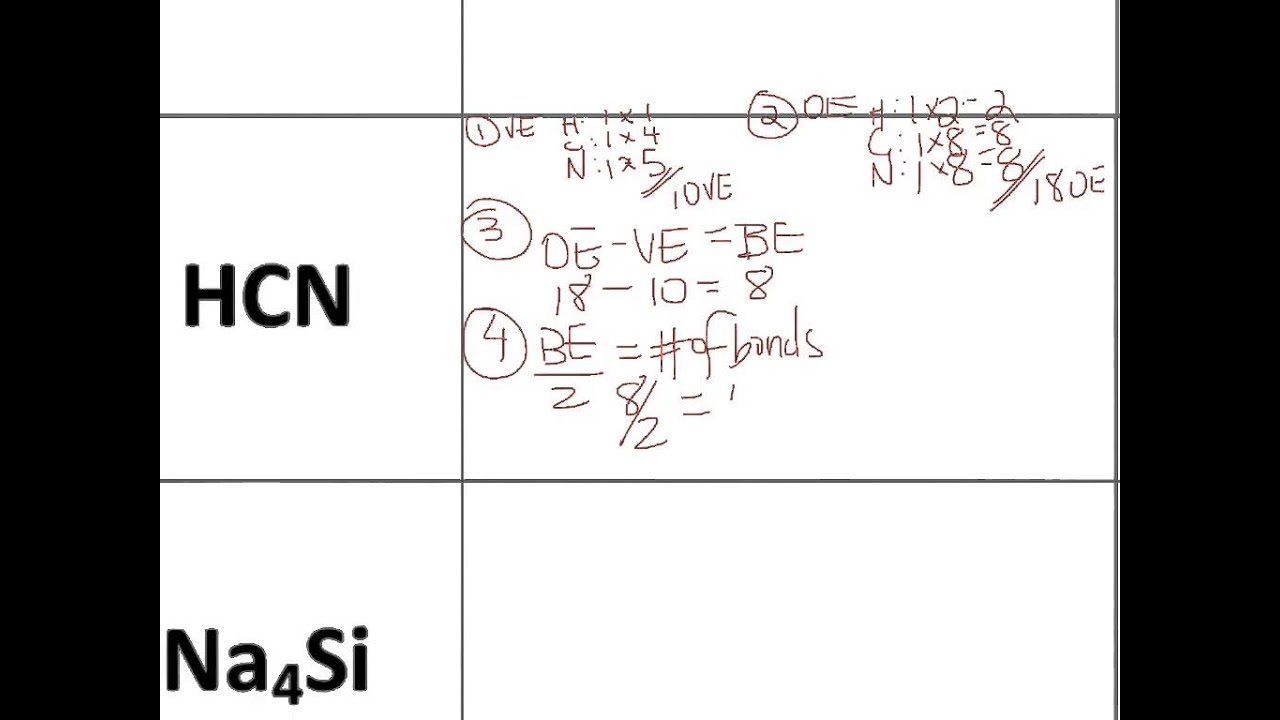
Chlorite is used in the paper, pulp, and bleaching of textiles. Chlorite has a +3 oxidation state and is part of the chlorine oxides family. In this article, we will study Chlorite ion (ClO2-) lewis structure, molecular geometry, hybridization, polar or nonpolar, bond angle, etc. There are two lone pairs of electrons on the central atom that do not participate in forming any bonds. Chlorite is one of the strongest oxidizers of chlorine oxyanions. The Lewis Structure of OF2 will have single bonds between O-F, with Oxygen atom in the centre. However, Oxygen atom is left with two pairs of nonbonding pairs of electrons. Every single bond will use up to 2 electrons.įluorine atoms will complete their octet by sharing electrons as now they will have eight electrons in their outer shell. Now each Fluorine atom forms a single bond with an Oxygen atom by sharing electrons. For example, Oxygen has six valence electrons, so draw six dots around the atom. Once you have placed all the atoms in this manner, start putting the valence electrons of the individual atom around them. As Oxygen is less electronegative than the fluorine atom, it will be placed in the centre. Chemical Lewis Structure Chemical Lewis Structure Formula Formula HCN CIF: NH3 SF2. In OF2, the Oxygen atom will acquire the central position, and both the Fluorine atoms will go on the sides like this. However, there are many exceptions to this rule, but we always keep this rule in mind while drawing the Lewis structure for any molecule. Remember that all atoms follow the octet rule, which mentions that an atom that wants to attain a stable structure like inert gases must have eight valence electrons in its outer shell. An explanation of the molecular geometry for the HCN ion (Hydrogen cyanide) including a description of the HCN bond angles. Now that we know the total number of valence electrons for OF2, we can make the Lewis dot structure of OF2. So here we will first look at the Lewis Structure of Oxygen Difluoride and then check out other properties of this molecule. This structure helps in determining other properties of the molecule. The Lewis structure of any molecule is a pictorial representation of the arrangement of valence electrons around individual atoms along with the bonds they form. Name of molecule Oxygen Difluoride ( O F 2 ) No of Valence Electrons in the molecule 20 Hybridization of O F 2 sp3 hybridization Bond Angles 109° 27′ Molecular Geometry of O F 2 Bentįor finding out the molecular geometry and hybridization of the molecule, it is vital first to know the Lewis Structure of OF2. It’s easy to understand the Lewis structure of this molecule as it has only two types of atoms in the molecule. This molecule’s chemical formula is OF2 as it contains one atom of Oxygen and two atoms of Fluorine. The angle between the C 2 axis of the SO 2 subunit and the NCH axis is 86° and the two axes intersect at a distance of 1.38 Å above the S nucleus.Oxygen Difluoride was first reported in 1929 while carrying out electrolysis of potassium chloride with hydrofluoric acid in the presence of water in little quantities. The distance between the SO 2 and HCN centers of mass is r c.m. 
The HCN molecule is roughly perpendicular to the plane of the SO 2 molecule with N lying between the SO 2 plane and the H atom. The spectroscopic constants have been interpreted in terms of a nonplanar, antihydrogen‐bonded geometry of C s symmetry with S, H, C, and N nuclei lying in the symmetry plane. Click on the image above to be taken to the WebMO. Predict the electron-pair geometry and molecular geometry of the XeF 4 molecule. The ground‐state rotational spectra of the four isotopic species ( 3 2SO 2,HC 1 4N), ( 3 2SO 2,DC 1 4N), ( 3 2SO 2,HC 1 5N), and ( 3 4SO 2,HC 1 4N) of a weakly bound dimer of sulfur dioxide and hydrogen cyanide have been observed by means of pulsed‐nozzle, Fourier‐transform microwave spectroscopy. Predicting Electron-pair Geometry and Molecular Geometry: XeF 4 Of all the noble gases, xenon is the most reactive, frequently reacting with elements such as oxygen and fluorine.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
